
Then comes oxidation number (hypothetical charge), which seems to be similar to electrovalency (actual charge), but still some online sources say these are different things. Now when I see carbon monoxide (CO), I am not able to figure out valency of. This is necessary because Co is a transition metal (d block element) and we need to take. Co-ordination compounds of Nira have been prepared by Jensen (1936) and Nyholm (1950), and the co-valency of 3 has been added by us. Carbon Dioxide Cycle and Formula How Carbon Dioxide is Produced The predetermined elemental molecule where valency element is zero, regardless of ionic compounds or covalent compounds, n-valence anion algebra and its composition are zero. Carbon - Element information, properties and uses Periodic Table What is. Now when I see carbon monoxide (CO), I am not able to figure out valency of carbon or oxygen. To find the number of valence electrons for Cobalt (Co) we need to look at its electron configuration. Bonds are formed between elements when they share or exchange one or more electrons. 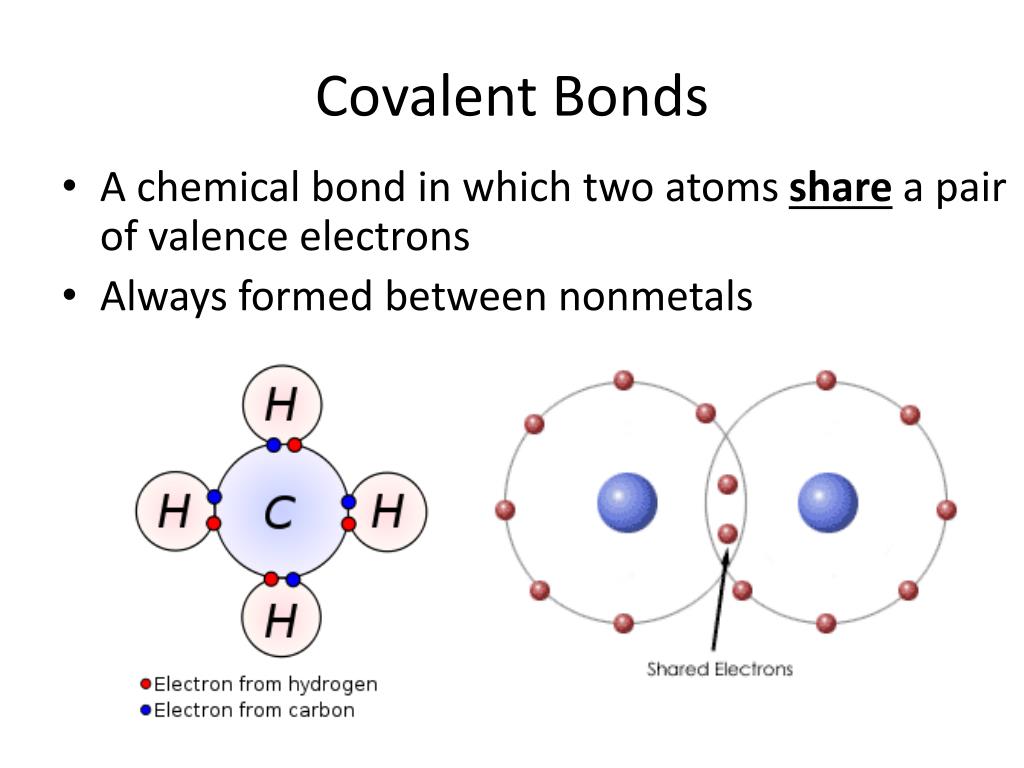
cobalt primary valency answer Valences of the Elements Chemistry Table. 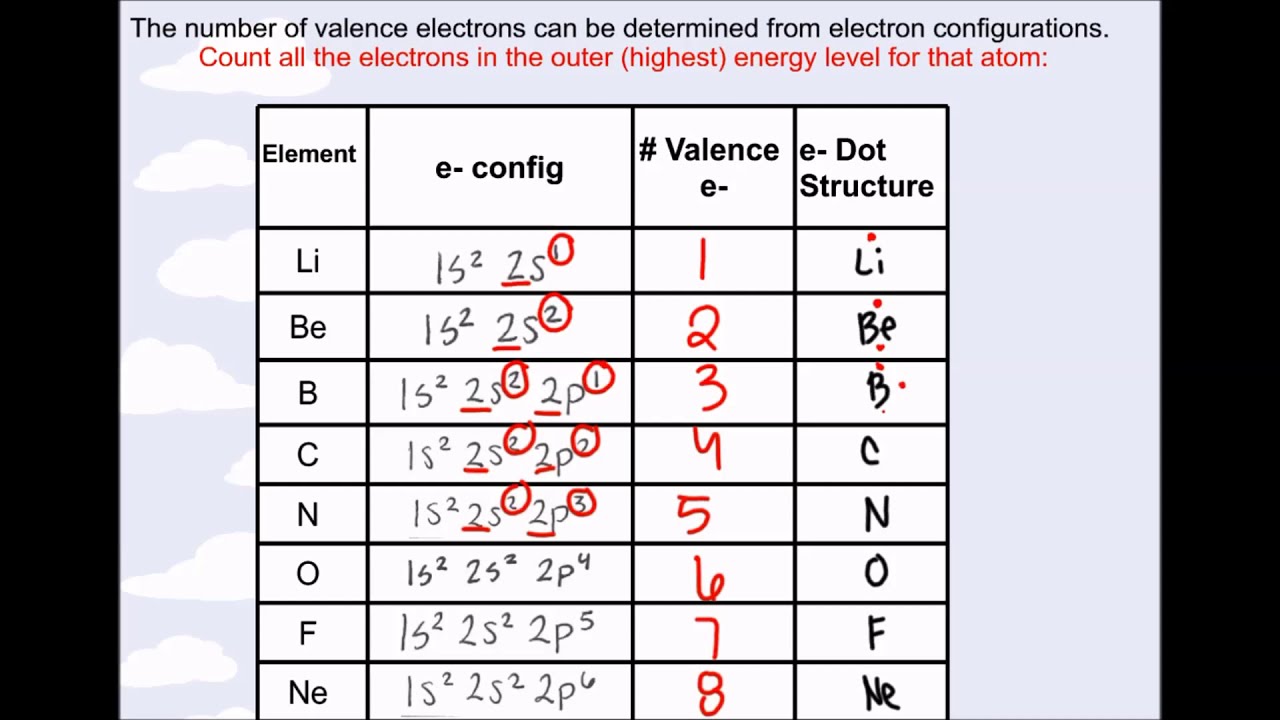
Should valency of carbon be 2 or 3 or 4?ģ- Electrovalency = number of charge on an ion.Ĥ- Covalency = number of shared electrons.ĥ- For an element covalency and electrovalency are equal. The valence state describes how many bonds can be made with the atom. It is an octahedral complex so secondary valency 6 or Co-ordination number 6. Super Trick to Find Covalency How to Find Covalenc圜ovalency is an important concept of Chemistry to learn and understand as it helps us many ways. About valency books say following things:ġ- Valency is combining capacity of an atom with number of hydrogen atoms or double of oxygen atoms with which the atom combines.ĭoubt A: what is valency of C in C2H2 and CO?Ģ- Valency is number of electrons gained, lost or shared by atom to complete its octate.ĭoubts B: in CO carbon shares 2 electrons and gains 2 electrons.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
